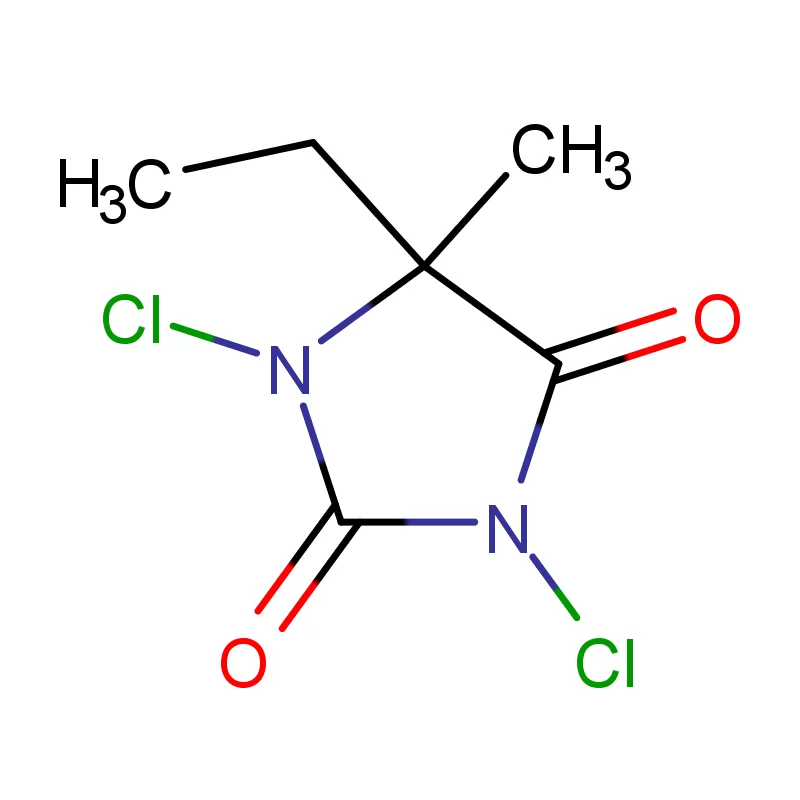
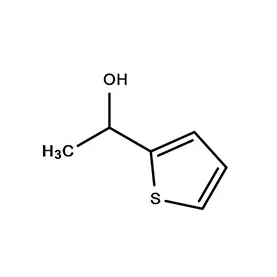
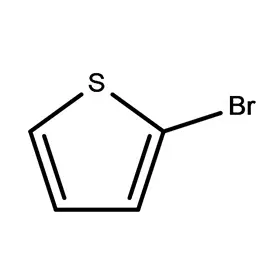
Uganda’s pharmaceutical industry is currently experiencing significant growth, driven by a rising population, increasing healthcare awareness, and government initiatives to promote local manufacturing. However, the industry faces challenges such as reliance on imported pharmaceutical intermediates, limited access to finance, and a shortage of skilled personnel. The tropical climate and agricultural base also influence the demand for specific fine chemicals used in pharmaceutical production.
The regulatory landscape is evolving, with increased scrutiny on product quality and safety. This is pushing manufacturers to adopt international standards and invest in advanced technologies. The demand for affordable medicines is high, creating a need for cost-effective 2 Bromothiophene sourcing and efficient production processes. Maintaining stable supply chains is also a key concern given Uganda’s landlocked location.
Despite these challenges, Uganda presents a promising market for pharmaceutical manufacturers. The government's commitment to reducing dependence on imports and fostering local production, combined with a growing middle class, creates a favorable environment for investment and innovation. Specifically, increasing needs for water purification in pharmaceutical manufacturing drives demand for industrial water treatment chemicals.