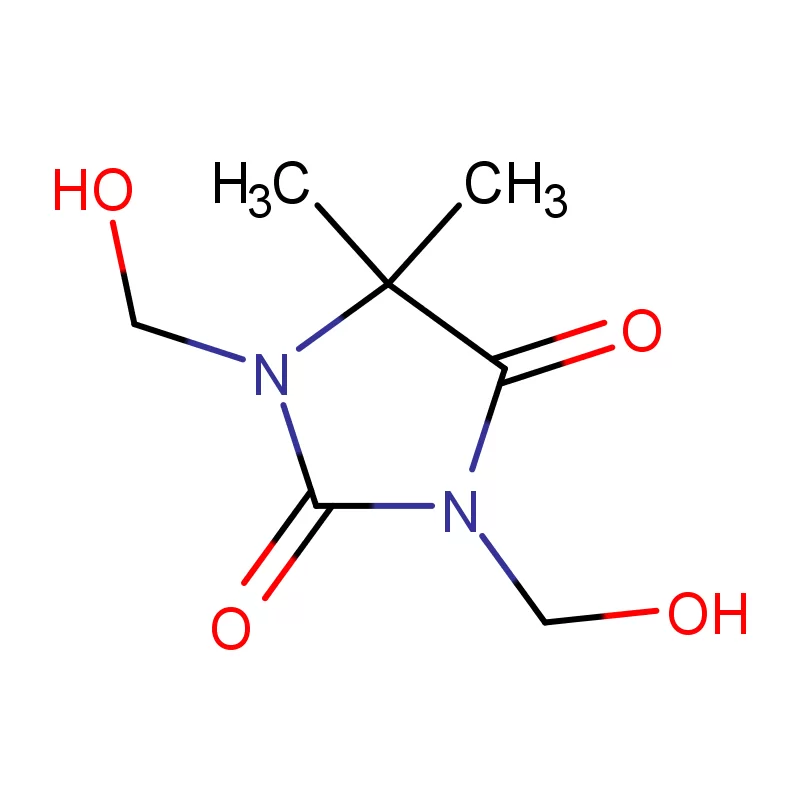
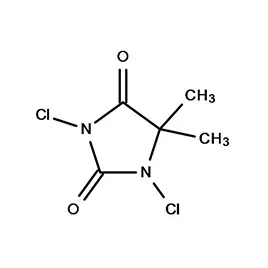
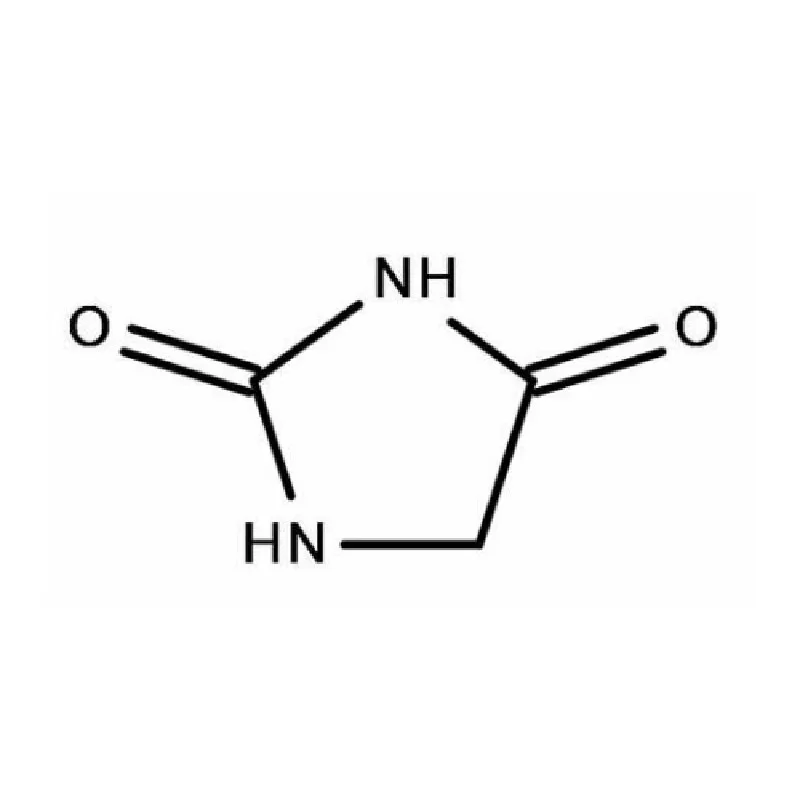
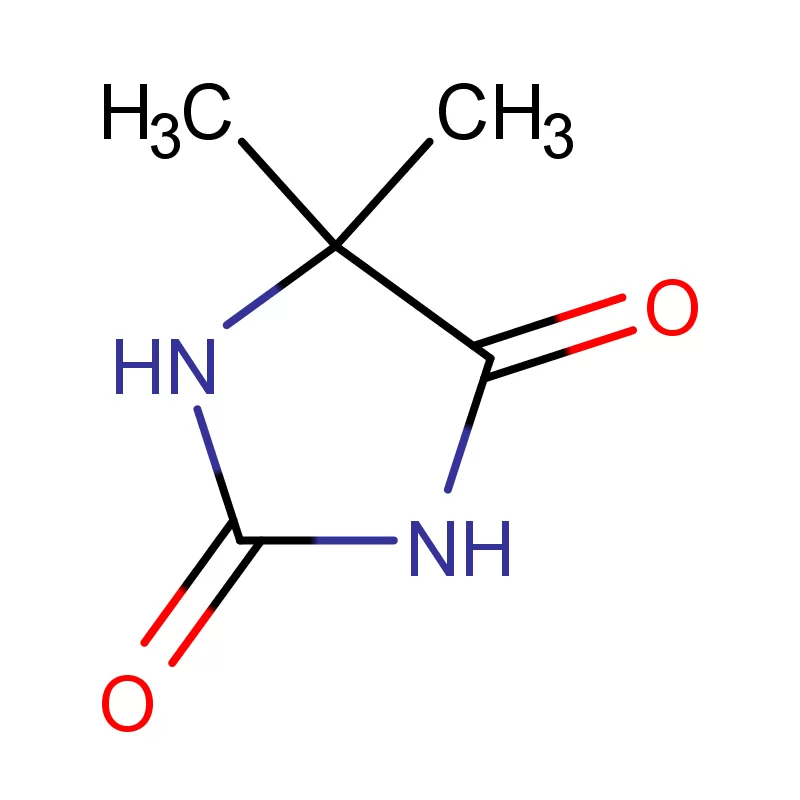
Bangladesh’s pharmaceutical sector has experienced significant growth in recent decades, driven by increasing healthcare access and a supportive government policy. The country is now a substantial producer of pharmaceutical intermediates and generic drugs, serving both domestic and international markets. However, challenges remain including reliance on imported raw materials like fine chemicals, and the need for greater investment in research and development.
The monsoon climate and frequent flooding in Bangladesh pose logistical challenges for the transportation and storage of sensitive chemical materials. Maintaining consistent quality control and ensuring adherence to international standards such as GMP are critical for companies operating within the country. The availability of skilled labor is a key asset, but continuous training and upskilling are vital to maintain competitiveness.
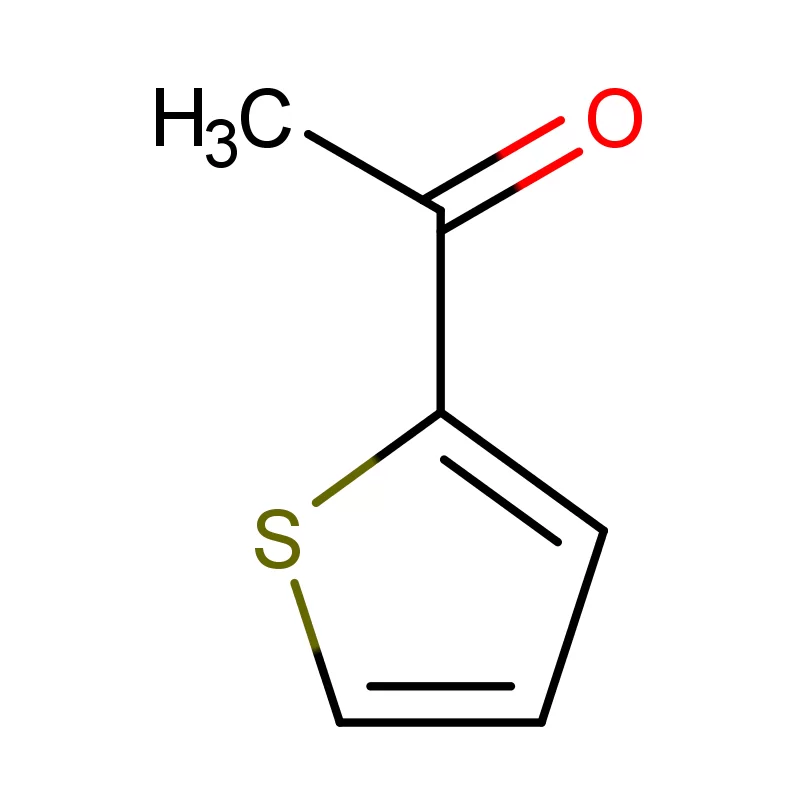
The local pharmaceutical industry is heavily reliant on a few key 2 Bromothiophene suppliers. This creates supply chain vulnerabilities. Increased demand for sustainable and eco-friendly chemical processes is also a growing trend driven by environmental concerns and regulatory changes. Effective industrial water treatment chemicals are essential for minimizing the environmental impact of pharmaceutical manufacturing.