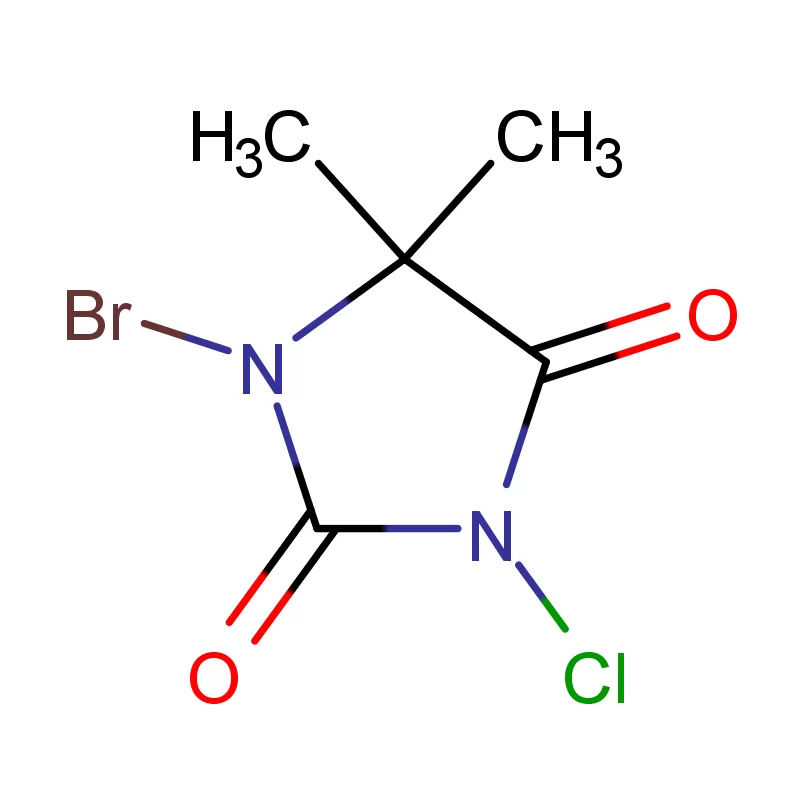
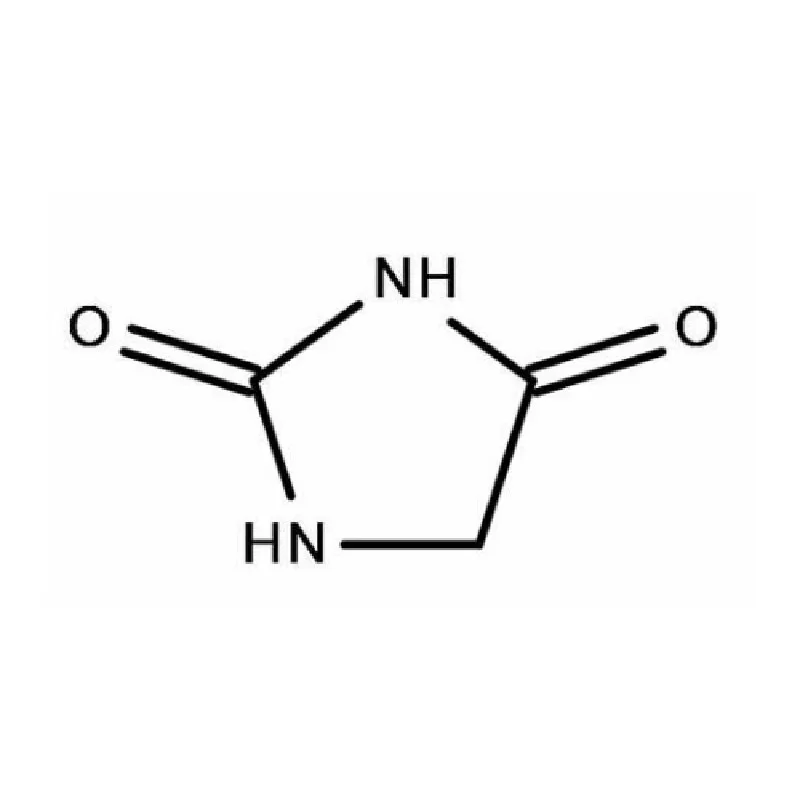
Uruguay's pharmaceutical industry is steadily growing, driven by an aging population and increasing healthcare investment. The demand for high-quality pharmaceutical intermediates is on the rise, but local manufacturing capacity is limited, creating reliance on imports. The country's stable political environment and robust regulatory framework attract foreign investment in the sector.
The Uruguayan climate, characterized by temperate conditions and relatively high humidity, presents specific challenges for chemical storage and handling. Manufacturers must invest in specialized infrastructure to maintain product integrity and prevent degradation. Furthermore, the geographical location of Uruguay influences logistical considerations, adding to transportation costs and lead times.
Despite these challenges, Uruguay offers a favorable business environment with a skilled workforce and a commitment to innovation. The government actively promotes the development of the pharmaceutical sector through incentives and support programs, fostering a dynamic and competitive market for fine chemicals.